Duchenne muscular dystrophy pedigree: inheritance, carrier detection and germline mosaicism
A clinical guide to reading, constructing, and counselling from a Duchenne muscular dystrophy pedigree — X-linked recessive inheritance, carrier mothers, de novo variants, and the maternal germline mosaicism that sits beneath many "sporadic" DMD cases.
Short version. Duchenne muscular dystrophy (DMD; ICD-10 G71.0; OMIM 310200) is an X-linked recessive dystrophinopathy caused by pathogenic variants in the DMD gene at Xp21.2. The classical pedigree shows affected males transmitted through carrier females across generations with no male-to-male transmission. Approximately one third of cases arise as de novo variants, but a clinically important fraction of those apparent de novo cases reflect maternal germline mosaicism, which is why sibling recurrence after a single affected boy is usually counselled at around 4% rather than at background. When a mother has two affected sons, the posterior probability of germline mosaicism rises sharply and recurrence risk becomes materially higher than the simple de-novo prior would suggest.
Clinical overview
Duchenne muscular dystrophy is the most common severe childhood muscular dystrophy, affecting roughly 1 in 3,500 live male births. Onset is typically between 2 and 5 years of age with proximal lower-limb weakness, calf pseudohypertrophy, Gowers' sign, and a waddling gait. Creatine kinase is markedly elevated from birth. Loss of ambulation is classical in the second decade without treatment, and cardiomyopathy and respiratory failure dominate the adult course. Female carriers are usually clinically unaffected but may have mildly elevated creatine kinase, subclinical cardiac involvement, or, in a minority with skewed X-inactivation, manifest muscle disease.
The allelic milder disorder, Becker muscular dystrophy, shares the same gene and inheritance but typically arises from in-frame DMD variants that preserve partial dystrophin function. For pedigree purposes, the two conditions are often drawn and annotated together, but their natural history, age of loss of ambulation, and cardiac trajectory differ meaningfully and should be captured in the catalogue entry.
Disease-modifying therapies now available include exon-skipping antisense oligonucleotides (eteplirsen for exon 51-amenable variants, golodirsen and viltolarsen for exon 53-amenable variants, casimersen for exon 45-amenable variants) and corticosteroids (prednisone, deflazacort, vamorolone). These treatments do not alter inheritance or recurrence risk, but they increase the clinical value of timely molecular diagnosis and of cascade screening across the pedigree, because eligibility for specific therapies depends on the exact DMD variant identified in the proband.
X-linked recessive inheritance in the pedigree
DMD follows classic X-linked recessive transmission. A hemizygous affected male transmits his X chromosome to all of his daughters, who become obligate carriers, and none of his sons. A heterozygous carrier female transmits the variant X to half of her offspring of either sex: on average half of sons are affected, half of daughters are carriers. The absolute pedigree fingerprint of X-linked recessive disease is the absence of male-to-male transmission: an affected father and an affected son, related through the father's Y chromosome, cannot both inherit the same X-linked variant by descent.
Read across generations, the classical DMD pedigree therefore shows:
- Affected males clustered across sibships related through unaffected mothers.
- Obligate carrier females where the pedigree structure demands it (for example, the mother and maternal grandmother of an affected boy whose maternal uncle is also affected).
- No transmission from affected father to affected son.
- Occasional manifesting female carriers, typically with milder phenotype and later onset, reflecting skewed X-inactivation.
For a deeper treatment of the calculation behind carrier and offspring probabilities, see our X-linked recessive calculator guide and the broader Mendelian inheritance calculator.
The maternal germline mosaicism story
Approximately one third of DMD cases are reported as de novo — that is, the pathogenic DMD variant was not detected in the mother's somatic DNA. If this were the whole story, sibling recurrence after a single apparently de novo affected boy would be close to the population background. In practice, counsellors quote approximately 4% sibling recurrence after a first apparently de novo DMD case. The gap between "0%" and "4%" is explained by maternal germline mosaicism.
Germline mosaicism arises when a mutation occurs in a precursor cell of the oocyte lineage after fertilisation of the mother. The mother herself is phenotypically normal and her blood DNA may show no detectable variant, or a very low-level variant well below heterozygous expectation (VAF near 50%). Her germline, however, contains a fraction of oocytes carrying the pathogenic DMD allele, and each of those oocytes has a 50% chance of transmitting it. DMD is one of the empirically best-characterised disorders for maternal germline mosaicism.
The counselling consequences are sharp. A single affected boy is compatible with either a true de novo event in the proband's meiosis or low-level maternal germline mosaicism; the two hypotheses cannot be separated by blood testing alone unless the mother's variant allele fraction is measurable. When a mother has two or more affected sons with the same DMD variant, the posterior probability of germline mosaicism rises dramatically — two independent de novo events in the same woman's meioses would be extraordinarily unlikely, whereas one germline-mosaic mother elegantly explains the observation. The practical outcome is that recurrence risk after the second affected son is materially above the ~4% empirical figure used after the first.
A pedigree example
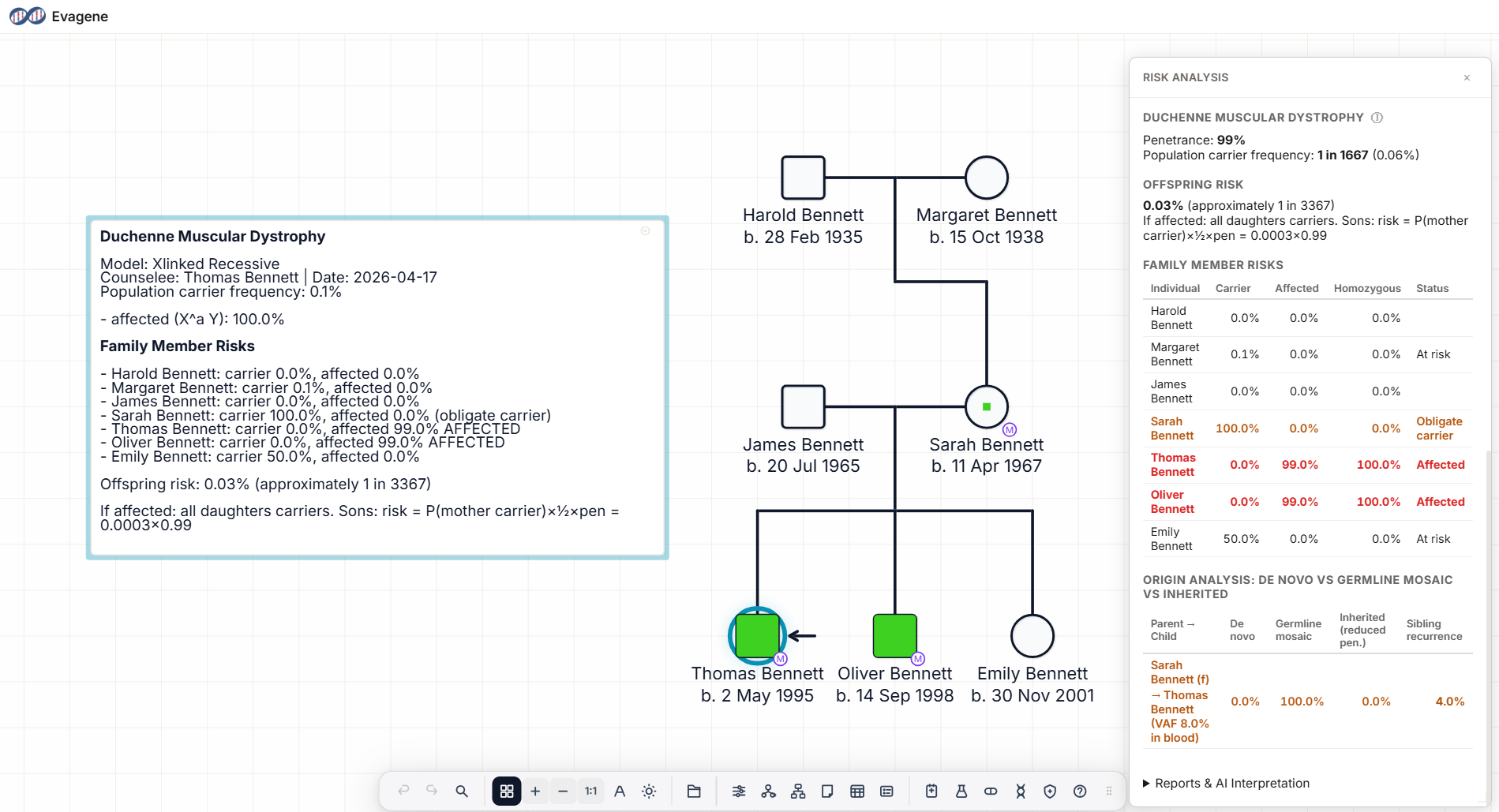
The pedigree above shows the scenario at the heart of DMD counselling. An unaffected mother and an unaffected father have three children: two affected sons, both with confirmed pathogenic DMD variants at the same genomic position, and one unaffected daughter. Targeted deep sequencing of the mother's peripheral blood reveals the same DMD variant at an allele fraction of 8%, far below the 50% expected for a heterozygous carrier and consistent with somatic mosaicism.
Three facts drive the analysis. First, two affected sons with the same variant from an unaffected mother is much better explained by germline mosaicism than by two independent de novo events. Second, a measurable blood VAF of 8% is direct evidence of mosaicism somewhere in the mother, and the germline fraction may be higher, lower, or similar to the blood fraction depending on when in development the mutation arose. Third, the daughter's status is not yet determined; she may be a non-carrier, a somatic-only mosaic, or a heterozygous-like carrier depending on which oocyte gave rise to her.
Reading the pedigree
When reading a DMD pedigree in clinic, work outward from the proband:
- Confirm the molecular diagnosis in the proband. DMD multi-exon deletions and duplications dominate the mutation spectrum; point mutations are a minority. The exact variant drives eligibility for exon-skipping therapy and guides cascade testing.
- Test the mother for the same variant at appropriate sensitivity. Targeted deep sequencing on blood is needed to detect low-level somatic mosaicism; a negative standard-sensitivity result on blood does not exclude germline mosaicism.
- Enumerate at-risk females: maternal sisters, maternal aunts, and their female descendants. In a true carrier pedigree, cascade testing identifies women at reproductive risk.
- Enumerate at-risk males: maternal brothers, maternal uncles, and their sons. In a carrier pedigree, half of sons of carriers are affected; early identification supports early initiation of corticosteroids and disease-modifying therapy.
- Record phenotypic nuance in female relatives: creatine kinase elevation, muscle symptoms, and cardiac findings in obligate or possible carriers are clinically important and should be annotated on the pedigree, not only in the clinic letter.
Recurrence risk counselling
Recurrence risk in DMD is calculated from three inputs: the prior probability that the mother is a constitutional heterozygous carrier, the prior probability that she is germline-mosaic, and any direct measurement of her variant allele fraction in blood or other accessible tissue. The arithmetic in outline:
- Mother confirmed heterozygous carrier (full-dose variant, blood VAF near 50%): recurrence risk for sons is approximately 50% affected; daughters approximately 50% carriers.
- Apparently de novo proband, mother blood-negative, single affected son: empirical sibling recurrence approximately 4%, driven by unobservable low-level germline mosaicism.
- Two or more affected sons, mother blood-negative or low VAF: posterior probability of germline mosaicism rises sharply; recurrence risk is best reported as a Bayesian posterior that combines the pedigree structure with any measured VAF, rather than as a fixed population figure.
- Mother with measured somatic VAF (e.g. 8%): recurrence risk approximates blood VAF × 0.5 for the germline fraction estimate, adjusted by the joint posterior incorporating the pedigree structure. The germline fraction may differ from the somatic fraction, and the uncertainty should be transmitted to the family.
In practice, the single most useful clinical habit is to record the number and sex of affected offspring, the number and sex of unaffected offspring, the results of targeted deep sequencing in the mother, and the measured VAF (if any) directly on the pedigree, so that the posterior calculation is transparent and reproducible.
How Evagene supports Duchenne muscular dystrophy pedigrees
Evagene is a browser-based pedigree platform in which Duchenne muscular dystrophy is a first-class catalogue entry. The DMD entry carries ICD-10 G71.0, OMIM 310200, X-linked recessive inheritance, a de novo rate prior of approximately 0.33, and condition-specific germline mosaicism parameters with a maternal sex bias. Drawing a pedigree and annotating the proband with DMD automatically makes all of these parameters available to the analysis surface; the clinician does not need to re-enter them from the literature.
The X-linked recessive carrier probability calculator runs directly on the pedigree to produce per-individual carrier and affection probabilities, taking into account the observed pedigree structure. The germline mosaicism posterior accepts the pedigree structure, optional blood VAF, and optional sperm VAF, and returns a calibrated joint posterior for each parent — the critical calculation when counselling families with two or more affected sons or a measurable maternal blood VAF. Evagene's batch screening surface, described in our batch pedigree risk screening guide, flags DMD alongside the other 200-plus catalogued conditions when the pedigree structure crosses relevant thresholds.
On top of the numerical analysis, Evagene offers AI-assisted clinical interpretation using bring-your-own-key Anthropic Claude or OpenAI GPT models. For a DMD pedigree with maternal mosaicism, the interpretation output covers the inheritance pattern, the germline mosaicism posterior with its assumptions, the cascade testing plan for at-risk females and males, and the data gaps (for example, unsequenced maternal relatives) that should be closed before the next clinic visit. The pedigree, its annotations, and the report are all exportable as GEDCOM, JSON, PDF, and through the REST API and MCP server for downstream systems.
Frequently asked questions
What is the inheritance pattern of Duchenne muscular dystrophy?
DMD is X-linked recessive. The DMD gene sits on Xp21.2. Males are hemizygous and therefore affected; heterozygous females are usually carriers but can manifest due to skewed X-inactivation. The fingerprint is absence of male-to-male transmission.
How common are de novo DMD mutations?
Approximately one third of DMD cases are de novo — not inherited from a carrier mother. A clinically important fraction of those reflects maternal germline mosaicism rather than a truly sporadic event.
Why does DMD counselling need to consider germline mosaicism?
Because blood-negative mothers may still carry the variant in their oocyte lineage. Empirical sibling recurrence after a single apparently de novo DMD case is around 4%, reflecting residual maternal germline mosaicism rather than true background risk.
Can I detect a DMD carrier from the pedigree alone?
A pedigree with multiple affected males related through unaffected females strongly suggests carrier status. A single affected boy can be either a carrier pedigree or a de novo / mosaic case; molecular testing is required to separate them.
What does a blood VAF of 8% mean for a DMD mother?
It is direct evidence of somatic mosaicism. Combined with pedigree structure, it feeds into a posterior estimate of germline mosaicism and recurrence risk. The germline fraction may differ from the somatic fraction.
Do exon-skipping therapies change DMD recurrence risk?
No. They change disease natural history, not inheritance. They do make early diagnosis and cascade testing more consequential because eligibility is variant-specific.
How does Evagene handle DMD?
DMD is a first-class catalogue entry with ICD-10, OMIM, inheritance, and mosaicism parameters. The X-linked recessive calculator, germline mosaicism posterior, batch screening, and AI interpretation all run directly on the pedigree.