Achondroplasia pedigree: FGFR3 inheritance, paternal age effect and sperm VAF counselling
A clinical guide to reading and counselling from an achondroplasia pedigree — FGFR3 c.1138G>A autosomal dominance, the paternal age effect, and the use of measured sperm variant allele fraction as a direct input into recurrence risk calculation.
Short version. Achondroplasia (ICD-10 Q77.4; OMIM 100800) is the commonest form of disproportionate short-limbed short stature. Inheritance is autosomal dominant with penetrance approximately 99%. A single recurrent pathogenic variant in FGFR3, c.1138G>A (p.Gly380Arg), accounts for more than 98% of cases. Approximately 80% of cases are de novo. The paternal age effect — driven by selective expansion of spermatogonial stem cells carrying the activating variant — means the mutation arises almost exclusively on the paternal allele. Sperm sequencing for the father produces a measurable variant allele fraction (VAF), and recurrence risk can be calibrated directly as approximately sperm VAF × 0.5 × penetrance. When the father's sperm VAF is measurably positive, the joint-parent posterior concentrates on paternal origin and the mother can be exonerated from meaningful contribution to recurrence risk.
Achondroplasia: clinical overview
Achondroplasia is the commonest non-lethal skeletal dysplasia, with a birth prevalence of approximately 1 in 25,000 live births. Clinical features include disproportionate short stature with rhizomelic shortening of the limbs, macrocephaly with frontal bossing and midface hypoplasia, trident hand posture, thoracolumbar gibbus in infancy followed by exaggerated lumbar lordosis, foramen magnum stenosis, and small cuboid vertebral bodies with decreasing interpediculate distance moving caudally in the lumbar spine. Intelligence is typically normal.
Clinical surveillance across the lifespan focuses on foramen magnum stenosis and cervicomedullary compression (infancy), sleep-disordered breathing, otitis media and conductive hearing loss, spinal stenosis (adulthood), genu varum, and obesity. Endoscopic foramen magnum decompression is performed when cervicomedullary compression is symptomatic or severe on imaging; lumbar decompressive surgery is common in adulthood. Growth hormone therapy remains debated; vosoritide, a C-type natriuretic peptide analogue that antagonises FGFR3 downstream signalling, is approved for growth promotion in children with open epiphyses.
Adult height in achondroplasia is typically around 131 cm for men and 124 cm for women. Life expectancy is modestly reduced relative to the general population, principally by increased cardiovascular and neurological complications in adulthood.
FGFR3 genetics and the paternal age effect
FGFR3 encodes fibroblast growth factor receptor 3, a transmembrane tyrosine kinase that negatively regulates endochondral ossification. The canonical achondroplasia variant, FGFR3 c.1138G>A (p.Gly380Arg) within the transmembrane domain, is an activating gain-of-function change that increases constitutive receptor signalling and suppresses chondrocyte proliferation and differentiation. A minority of cases carry c.1138G>C, which produces the same p.Gly380Arg amino acid substitution. Allelic disorders include hypochondroplasia (milder phenotype, different FGFR3 variants), thanatophoric dysplasia types I and II (lethal at birth), SADDAN (severe achondroplasia with developmental delay and acanthosis nigricans), and Muenke syndrome (craniosynostosis).
The most striking feature of achondroplasia genetics is the mutational mechanism. The c.1138G>A variant is not simply a high-frequency de novo event driven by sequence context; it is a selected mutation in the male germline. Spermatogonial stem cells are a self-renewing population in the adult testis, dividing many times over a man's reproductive lifespan. A mutation arising in a single spermatogonium that confers a proliferative advantage will clonally expand over subsequent divisions, increasing the fraction of sperm carrying that variant with age. Activating variants in FGFR3 and several other growth-signalling receptors provide such an advantage — the mutant stem cell out-competes its neighbours.
The empirical consequence is the paternal age effect. The rate of de novo FGFR3 achondroplasia variants in sperm rises steeply with paternal age, which is why advanced paternal age is associated with increased birth prevalence of achondroplasia, and why the recurrent c.1138G>A mutation is the dominant mutational entry point. Approximately 80% of achondroplasia cases are de novo; the vast majority of those de novo events occur on the paternal allele.
Sperm VAF as a counselling input
The clinical relevance of the spermatogonial selection mechanism is that it produces a measurable fraction of mutant sperm. A man who has fathered an achondroplasia pregnancy can have his sperm tested by targeted deep sequencing or by droplet digital PCR for the FGFR3 c.1138G>A variant. The result is a variant allele fraction (VAF) — the proportion of sequencing reads carrying the variant — which estimates the proportion of his sperm carrying the variant.
Recurrence risk is then directly calibrated:
- Recurrence risk per conception ≈ sperm VAF × 0.5 × penetrance, where 0.5 reflects the fact that a sperm carrying the variant fertilises approximately half of conceptions, and penetrance for achondroplasia is approximately 0.99.
- Example: sperm VAF 12%, penetrance 0.99, recurrence per conception ≈ 12% × 0.5 × 0.99 ≈ 5.9%.
- Example: sperm VAF below detection limit at adequate sensitivity, penetrance 0.99, recurrence per conception scales with the detection threshold of the assay and is typically well under 1%.
This is a qualitatively different counselling position from the population-prior de novo figure. Rather than quoting an empirical sibling recurrence of a few percent from the literature, the clinician has a direct, quantitative, family-specific number that can support prenatal, preimplantation, and partner-selection decisions.
A pedigree example
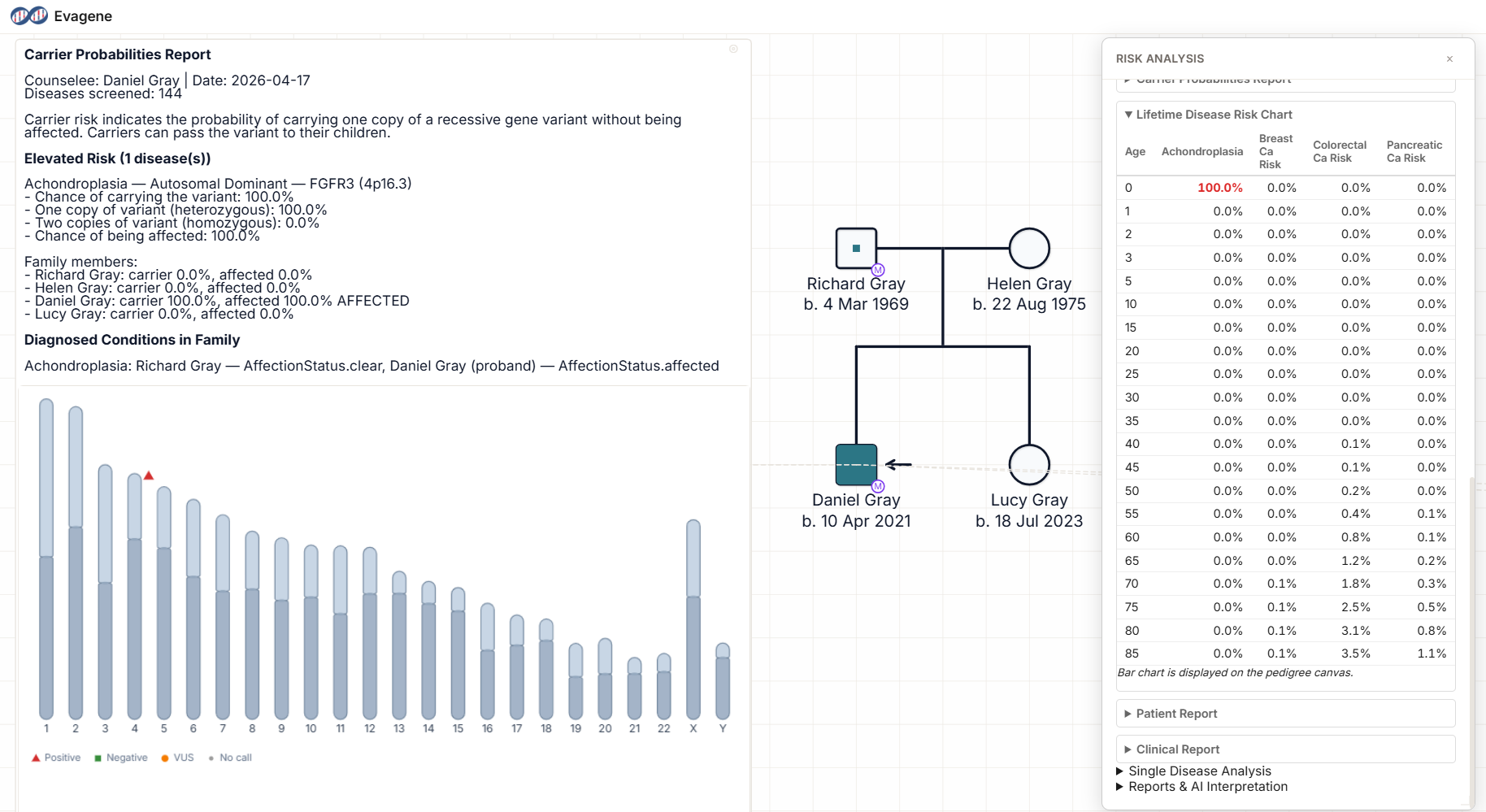
The pedigree above shows a practical achondroplasia counselling case. The father, of advanced paternal age, is phenotypically unaffected. The mother is unaffected. The couple has two children: an affected child with classical achondroplasia and a molecularly confirmed FGFR3 c.1138G>A variant, and an unaffected child. The father has undergone sperm sequencing for the variant, which returns a VAF of 12%. The mother's blood testing is negative at adequate sensitivity.
Three steps of interpretation:
- Joint-parent posterior. With a measurable paternal sperm VAF of 12% and a negative maternal blood test, the posterior probability that the variant arose in the father's germline is effectively 100%. The mother is exonerated.
- Recurrence estimate. Father's sperm VAF 12%, penetrance 0.99: recurrence per conception ≈ 12% × 0.5 × 0.99 ≈ 5.9%. This is a calibrated figure specific to this family.
- Other children. The unaffected child's risk of carrying the variant asymptomatically is very low at 0.99 penetrance; incomplete penetrance in achondroplasia is clinically minimal, so the unaffected child is usually counselled as non-carrier for practical purposes.
If the couple separates and the father re-partners, the recurrence risk is driven by his sperm VAF and does not depend on the new partner's history. Conversely, if the mother re-partners with a new biological father whose sperm is not mosaic for FGFR3, her recurrence risk for achondroplasia returns to the population background. This is a concrete illustration of the joint-parent logic: mosaicism attaches to a specific parent, not to the couple.
Recurrence risk from measured VAF and joint-parent logic
The joint-parent posterior in Evagene's germline mosaicism analysis handles the full set of scenarios that arise in achondroplasia counselling:
- Single affected child, both parents blood-negative, no sperm VAF: posterior probability of paternal germline mosaicism at its prior (~0.5%), recurrence driven mainly by the residual probability of an unobserved paternal germline event.
- Single affected child, measurable paternal sperm VAF: posterior paternal probability effectively 100%; recurrence calibrated directly from VAF.
- Single affected child, paternal sperm VAF below detection: posterior paternal probability reduced but not zero; recurrence scales with the detection threshold.
- Two affected children, same variant, unaffected parents: posterior concentrates on paternal germline mosaicism; recurrence estimate rises substantially.
- Affected father, heterozygous: recurrence 50% × penetrance per conception; sperm testing not needed for counselling.
- Both parents affected (homozygosity risk): see the homozygous achondroplasia discussion below.
Homozygous achondroplasia — two FGFR3 c.1138G>A variants in trans — is perinatally lethal, with severe thoracic restriction and pulmonary hypoplasia. In couples where both partners have heterozygous achondroplasia, each conception has 25% risk of homozygous lethal disease, 50% heterozygous achondroplasia, and 25% non-carrier. Prenatal and preimplantation testing are commonly offered.
For the underlying autosomal dominant arithmetic, see our autosomal dominant calculator. For the full Mendelian framework, our Mendelian inheritance calculator guide is the starting point.
How Evagene supports achondroplasia pedigrees
Achondroplasia is a first-class catalogue entry in Evagene. The record carries ICD-10 Q77.4, OMIM 100800, autosomal dominant inheritance, penetrance ~0.99, de novo rate ~0.8, germline mosaicism rate ~0.005, and a paternal sex bias that encodes the spermatogonial selection mechanism. The canonical variant, FGFR3 c.1138G>A (p.Gly380Arg), can be annotated directly on the proband so that downstream reports carry the correct molecular detail.
The germline mosaicism posterior calculator accepts optional blood VAF for each parent and optional sperm VAF for the father, and applies the paternal bias encoded in the catalogue. For the worked example above, a sperm VAF of 12% produces a calibrated recurrence estimate with transparent assumptions: the clinician can show the family exactly how the 5.9% figure was derived. The inheritance pattern identifier supports upstream analysis where the inheritance model is not yet confirmed.
AI-assisted clinical interpretation using bring-your-own-key Anthropic Claude or OpenAI GPT models generates a structured report covering FGFR3 inheritance, the paternal age effect, the sperm-VAF-calibrated recurrence estimate, the homozygous-risk discussion if relevant, prenatal and preimplantation testing options, vosoritide eligibility, and the clinical surveillance plan. The pedigree, reports, and molecular detail flow through the REST API and MCP server to downstream systems.
Frequently asked questions
What is the inheritance pattern of achondroplasia?
Autosomal dominant. The canonical variant is FGFR3 c.1138G>A (p.Gly380Arg), accounting for more than 98% of cases. Approximately 80% of cases are de novo.
Why is c.1138G>A the canonical variant?
Because it is positively selected in spermatogonial stem cells — the activating variant confers a proliferative advantage. Clones carrying it expand with paternal age, raising the fraction of mutant sperm.
What is the paternal age effect?
Older fathers' sperm carry an increased fraction of activating FGFR3 variants because mutant spermatogonial clones expand with age. This drives the age-dependent birth prevalence.
How is sperm VAF used in counselling?
Recurrence per conception ≈ sperm VAF × 0.5 × penetrance. A sperm VAF of 12% at penetrance 0.99 produces a recurrence of approximately 5.9%.
Can the mother be exonerated when the father is mosaic?
Yes. With a measurable paternal sperm VAF and negative maternal blood testing, the joint-parent posterior concentrates on paternal origin. The mother's contribution to recurrence is minimal.
What happens if two achondroplasia carriers have a child?
Each conception has 25% homozygous (perinatally lethal) achondroplasia, 50% heterozygous achondroplasia, and 25% non-carrier. Prenatal and preimplantation testing are commonly offered.
How does Evagene handle achondroplasia?
Achondroplasia is a first-class catalogue entry with OMIM, ICD-10, inheritance, penetrance, de novo rate, and paternally-biased germline mosaicism parameters. The joint-parent posterior accepts sperm VAF directly, and AI interpretation generates family-specific reports.