Germline mosaicism calculator: recurrence risk from pedigree structure, somatic VAF, and joint-parent logic
The clinical counselling problem that standard Mendelian risk models do badly: two affected children from unaffected parents. Is it de novo, reduced penetrance, or parental germline mosaicism — and what is the recurrence risk for the next pregnancy?
Short version. When an autosomal dominant or X-linked dominant condition appears in two or more children of phenotypically unaffected parents, the recurrence risk for the next pregnancy is neither the textbook "50% if inherited" nor the "zero if de novo" — it is a weighted mixture of hypotheses dominated by the probability that one parent is germline mosaic. Evagene computes this posterior directly from the pedigree structure, accepting optional somatic variant allele fraction (VAF) from blood or sperm sequencing as a sharpening input, and uses joint-parent logic so that confirming mosaicism in one parent properly exonerates the other. This page walks through the maths, the five textbook clinical scenarios (DMD, achondroplasia, osteogenesis imperfecta, tuberous sclerosis, Dravet), and the counselling implications.
Why germline mosaicism matters in clinical counselling
Consider a couple referred after the loss of two consecutive pregnancies to perinatal-lethal osteogenesis imperfecta type II. Both parents are clinically unaffected, both have negative blood sequencing for COL1A1 and COL1A2. The standard textbook answers fail here. It is not autosomal recessive OI: two de novo events in the same family would be astronomically unlikely. It is not a straightforward de novo dominant either, because the recurrence in a second pregnancy rules that out. What remains is parental germline mosaicism — one parent carrying the variant in a fraction of their gametes but not at detectable levels in their somatic cells.
This is not a rare edge case. It is the single most important reason a paediatric geneticist ever quotes "higher than de novo but lower than 50%" recurrence. Getting the number right matters because the counselling decision it drives — whether to pursue preimplantation genetic testing, invasive prenatal testing, or attempt another natural pregnancy — is a major life decision for the family. Quoting "zero because it was de novo" will be wrong often enough to cause harm; quoting "50% because someone must be a carrier" will be wrong too, usually in the direction of excessive anxiety and unwarranted invasive testing.
Traditional Mendelian risk calculators do not solve this problem. BRCAPRO, MMRpro, PancPRO are Bayesian models for specific cancer risk genes; they do not model germline mosaicism. A generic autosomal dominant inheritance calculator assumes heterozygous or homozygous parents, not mosaic parents. The result in practice is that germline mosaicism posterior reasoning is done on the back of the clinician's envelope — sometimes very well, often not. Evagene's contribution is to put the calculation on the same canvas as the pedigree, parameterised by the disease catalogue, and offered automatically whenever the pedigree pattern warrants it.
The model: what Evagene actually computes
Given a pedigree with an affected proband plus zero or more affected and unaffected siblings, two clinically unaffected parents, and a disease catalogue entry specifying inheritance_pattern, penetrance, de_novo_rate, germline_mosaicism_rate, and germline_mosaicism_sex_bias, Evagene computes:
- Posterior P(mother mosaic) — probability that the mother's germline carries the variant in some fraction f
- Posterior P(father mosaic) — likewise
- Posterior distribution over mosaic fraction f — where a run of affected children pushes the posterior toward higher f values
- Posterior P(true de novo) — variant arose in a single gamete, now present in the proband only
- Posterior P(inherited with reduced penetrance) — one parent silently heterozygous
- Recurrence risk for the next pregnancy — integrated across the above hypotheses
Each hypothesis starts from a prior set by the disease-catalogue parameters. For an AD disorder with de_novo_rate = 0.8 and germline_mosaicism_rate = 0.002, the priors for an isolated proband with unaffected parents are dominated by de novo. The likelihood of the observed pedigree under each hypothesis is then computed: a second affected child is a very low-likelihood event under the true de novo hypothesis (requires a second independent mutation, probability on the order of the per-gene mutation rate squared), but a plausible event under the germline mosaicism hypothesis (mosaic fraction f squared, times 0.5 squared). The posterior redistributes sharply toward mosaicism after the second affected child, and even more sharply after the third.
When somatic VAF is available, it enters as a direct likelihood on the mosaic fraction. A measured blood VAF of 8% in the mother of two affected DMD sons pins the mosaic-fraction posterior near 8% and makes the "mother mosaic" posterior numerically indistinguishable from 1.0 — at which point the "father mosaic" and "both mosaic" posteriors collapse to near zero under joint-parent logic.
Scenario 1: Duchenne muscular dystrophy with maternal blood VAF
A clinically unaffected mother has two affected sons and one unaffected daughter. Deep-coverage sequencing of maternal blood detects the family's DMD variant at 8% VAF. The pedigree looks like this:
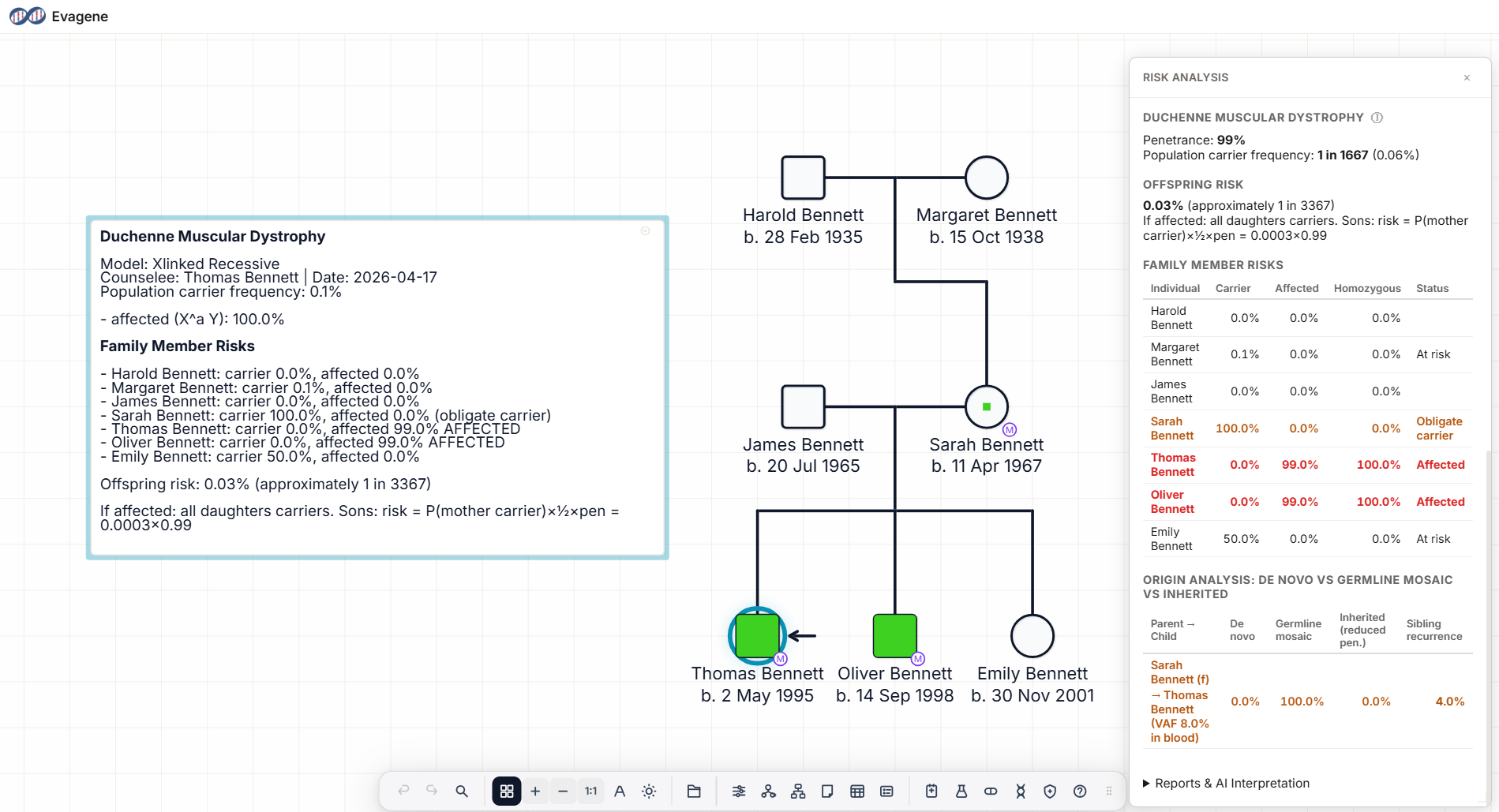
The maternal blood VAF of 8% anchors the posterior at near-certainty that the mother is germline mosaic with mosaic fraction ~8%. For an X-linked recessive condition, sibling recurrence risk for male offspring is approximately f × 0.5 × 1 (penetrance in hemizygous males) = ~4%, and for female offspring the risk of being a carrier is similarly ~4%. Male recurrence is well above the population DMD de novo rate of approximately 1 in 3,500 boys, but well below the 50% that would apply if the mother were confirmed heterozygous.
Counselling implications: prenatal testing is reasonable in all future pregnancies; preimplantation genetic testing is a sensible option for families pursuing further children; the unaffected daughter's carrier risk is ~4% and she should be offered testing before her own reproductive decisions. The Duchenne muscular dystrophy pedigree guide expands on the full workflow.
Scenario 2: Achondroplasia with measured sperm VAF and advanced paternal age
An advanced-paternal-age father has one affected and one unaffected child with achondroplasia. Sperm VAF for the canonical FGFR3 c.1138G>A variant is measured at 12%. The clinical question is whether the mother carries any residual risk and what the recurrence risk is for another pregnancy.
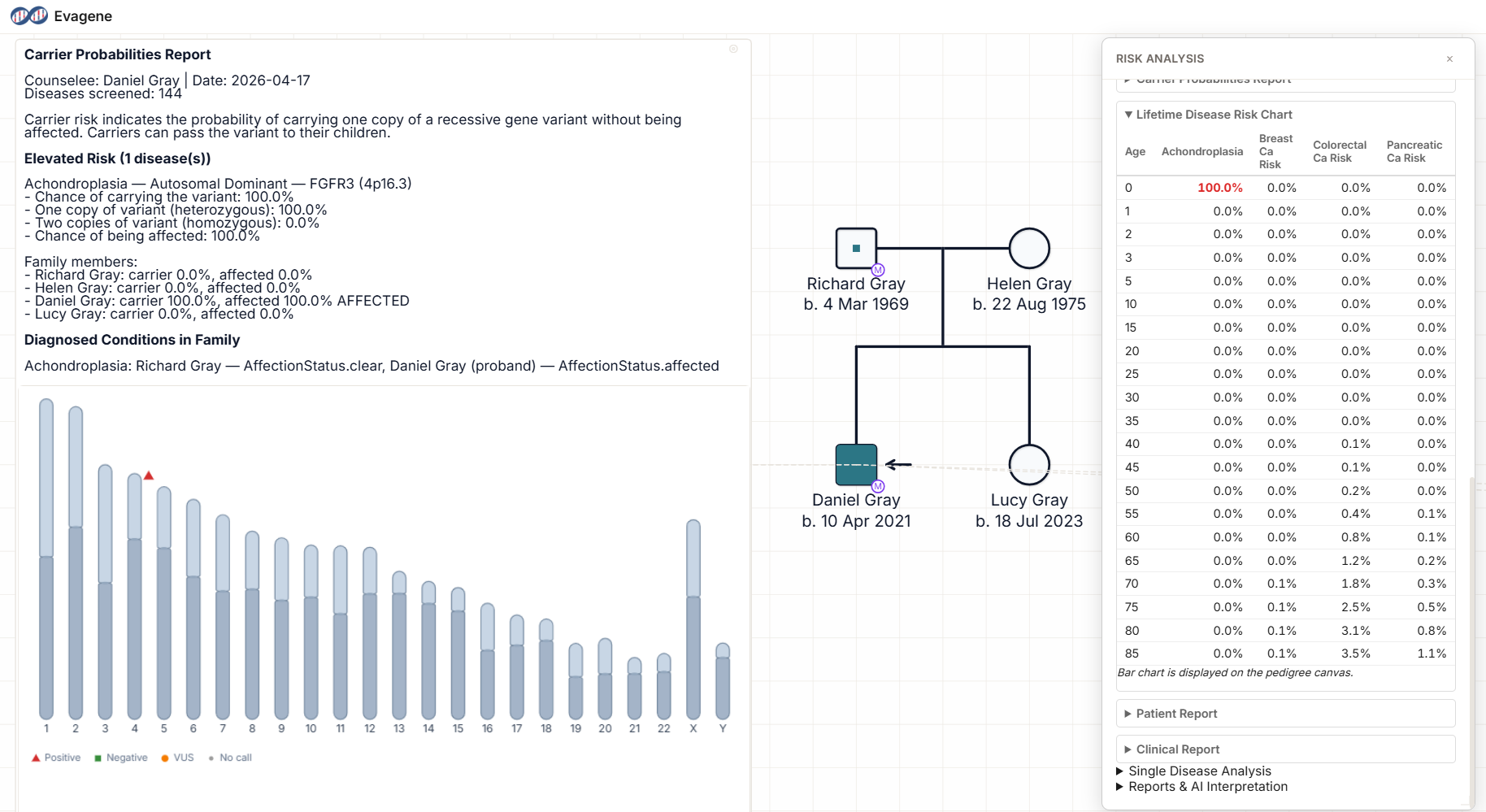
The 12% sperm VAF places the paternal mosaic posterior near 0.998. This is the kind of case where joint-parent logic matters most: without it, the maternal posterior would carry a small residual of co-suspicion because "a parent mosaic" was the best explanation of the family pattern even before the VAF test was run. With joint-parent logic, the maternal posterior collapses to near zero once the father's mosaicism is empirically confirmed. Mother is exonerated.
Recurrence risk for the next pregnancy is approximately sperm VAF × 0.5 × penetrance = 0.12 × 0.5 × 0.99 ≈ 6%. FGFR3 achondroplasia is the canonical paternal-age-effect disorder: the variant arises in spermatogonial stem cells that selectively expand, and sperm VAF rises with paternal age. See our achondroplasia pedigree guide for the full clinical picture.
Scenario 3: Osteogenesis imperfecta, two lethal neonates, no VAF
Two perinatal-lethal OI neonates from clinically unaffected parents with negative blood sequencing. A current pregnancy is underway. No VAF measurement is available.

Without VAF data, the calculation runs on pedigree structure alone. The disease-catalogue prior sets de_novo_rate ≈ 0.8 for perinatal-lethal OI type II and germline_mosaicism_rate ≈ 0.002. After two affected neonates, these priors are transformed dramatically: the true-de-novo hypothesis drops to the per-gene mutation-rate-squared order (effectively zero), and both parents' mosaic posteriors rise to approximately 99% each. Which parent is actually mosaic cannot be distinguished without gamete VAF. Recurrence risk for the current pregnancy integrates over both hypotheses and lands at approximately 13% — the classic number quoted in the OI counselling literature.
This is the single most important case for mosaicism-aware counselling. Without it, a clinician might tell the parents that the first lethal OI was "a de novo tragedy, unlikely to recur" — only for a second lethal pregnancy to reveal that the risk was always ~13%, not ~0%. The counselling failure is avoided by computing the mosaicism posterior at the first affected pregnancy, acknowledging that the number is small then (the prior is dominated by de novo), but logging it and being ready to update sharply at a second pregnancy. See the osteogenesis imperfecta pedigree guide.
Scenario 4: Tuberous sclerosis, three affected children, no VAF
Three of three children affected with tuberous sclerosis from clinically unaffected parents. No VAF data.
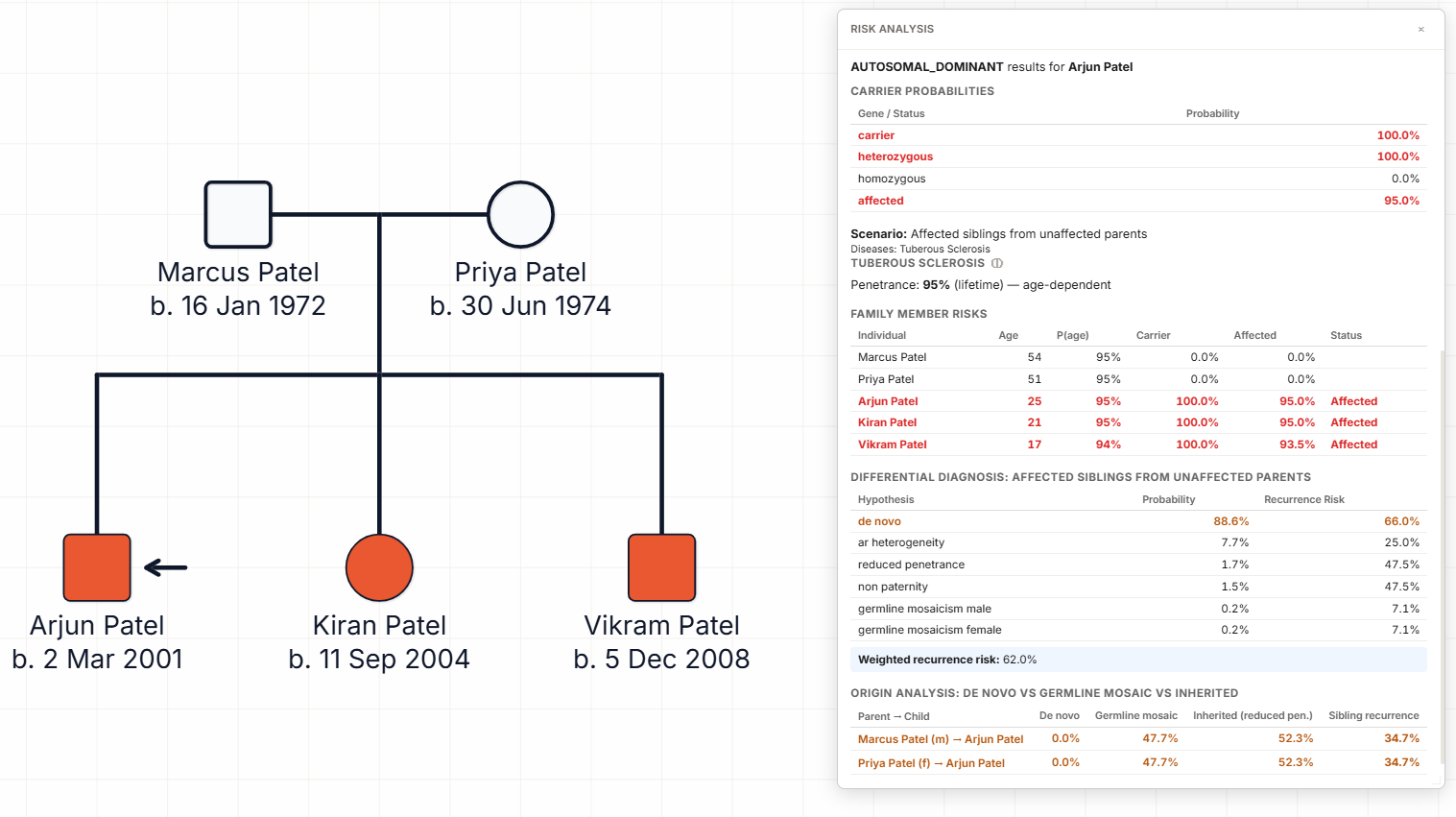
Three-of-three is a different arithmetic from two-of-two. Under the true-de-novo hypothesis, three independent events is essentially impossible. Under the mosaicism hypothesis, the likelihood of three affected children given mosaic fraction f is proportional to f³, which concentrates the posterior mosaic fraction on high values (> 30%). Under the inherited-with-reduced-penetrance hypothesis, TSC's known variable expressivity makes a silent heterozygous parent plausible if penetrance can be as low as ~0.5. The posterior splits roughly: mosaic ~48%, inherited reduced penetrance ~52%, with recurrence risk for a fourth pregnancy approximately 35%.
Note what the model is not doing: it is not collapsing to the mosaicism hypothesis just because the clinician thought of it. The reduced-penetrance hypothesis is a legitimate competitor when the disease catalogue admits it. The counselling message — "recurrence risk around 35%, testing the proband's variant directly in parental tissues is the most informative next step" — is the right message regardless of which of the two hypotheses is dominant. See the tuberous sclerosis pedigree guide.
Scenario 5: Dravet syndrome, isolated proband, no recurrence
An isolated Dravet syndrome proband with a confirmed de novo SCN1A pathogenic variant, two clinically unaffected siblings, unaffected parents. No VAF data. This is the far more common scenario, and the one where mosaicism-aware counselling matters in the other direction.
The disease catalogue prior for Dravet gives de_novo_rate ≈ 0.9 and germline_mosaicism_rate ≈ 0.07. With only one affected child and two unaffected siblings, the posterior stays dominated by true de novo: the "both siblings could have been unaffected by chance" likelihood is high under de novo (unity) and modest under low-fraction mosaicism (each unaffected sibling is unlikely under mosaic transmission). The calculation lands at approximately 99.9% de novo, residual recurrence risk less than 1%.
The correct counselling message here is: "This is overwhelmingly likely to be de novo. The residual recurrence risk is under 1%, dominated by the small but non-zero possibility of parental germline mosaicism for which we cannot rule out by negative blood sequencing. For reassurance, sperm VAF testing is available and would reduce the residual further; most families do not pursue it." The Dravet syndrome pedigree guide walks the full pattern.
Scenario 6: Rett syndrome — the other isolated-proband case
An isolated female proband with a de novo MECP2 variant. Unaffected siblings, unaffected parents. Rett syndrome is X-linked dominant with strong female bias (males usually lethal in utero or very severe). Catalogue priors: de_novo_rate ≈ 0.99, germline_mosaicism_rate ≈ 0.005, paternal bias.
Rett is the inverse of Dravet — higher de novo rate, lower mosaicism rate, and a pronounced paternal origin pattern where the variant arises in the father's sperm. Posterior: ~100% de novo, recurrence ~0 for future pregnancies (mothers can have additional affected children only via the rare maternal germline mosaicism, and fathers do not normally transmit because the paternal new mutation is a one-off). Counselling reassurance is very strong. See the Rett syndrome pedigree guide.
Somatic VAF as a clinical input
Somatic variant allele fraction from blood, saliva, sperm, buccal swab, or solid tissue sequencing is increasingly available as testing depth increases. Evagene accepts VAF annotations on any individual in the pedigree, tagged to the tissue of origin and the specific test. The calculator uses VAF in two ways:
- As direct evidence of mosaicism. A positive VAF in a phenotypically unaffected parent is direct confirmation of mosaicism in that tissue. Sperm VAF is the gold standard for paternal germline mosaicism; blood VAF is a lower-bound estimate for maternal germline mosaicism (germline and blood lineages diverge early in embryogenesis, so blood VAF may under-represent germline fraction).
- As a recurrence-risk anchor. For AD disorders, recurrence risk ≈ VAF × 0.5 × penetrance once the mosaic parent is identified. For XLR disorders, the maternal-mosaic contribution to male recurrence is similarly VAF × 0.5 × 1.
Absence of VAF data does not paralyse the calculation — the prior plus pedigree likelihood produces an informative posterior — but adding VAF meaningfully sharpens it, especially in families where a second affected pregnancy has raised the mosaicism posterior to ambiguous levels.
Joint-parent logic: why it matters
Naive implementations estimate each parent's mosaic posterior independently from the same pedigree evidence and present both numbers to the clinician. This is statistically incorrect: the events "mother is mosaic" and "father is mosaic" are mutually exclusive in almost all realistic scenarios, so finding one of them to be very likely should push the other toward zero, not leave both at ~99%.
Joint-parent logic in Evagene models the hypotheses as a mutually exclusive set: {mother mosaic, father mosaic, inherited with reduced penetrance, true de novo, technical artefact}. The posterior over this set sums to 1. Any evidence that raises one hypothesis (a positive paternal sperm VAF, a maternal blood VAF, a historical family-history clue) properly lowers the others. The clinical benefit is clean counselling: "father confirmed mosaic, mother's risk of being mosaic is now effectively zero, recurrence risk comes solely from paternal gamete VAF."
Using the calculator in Evagene
The mosaicism posterior is not a separate calculator — it is integrated into the pedigree risk engine. Construct the pedigree on the canvas, annotate affected status and age of onset, assign the disease from the catalogue (de_novo_rate and germline_mosaicism_rate are already populated), and optionally add VAF measurements on any individual's sequencing record. Running risk analysis produces the mosaicism posterior alongside carrier probabilities and future-risk estimates, with recurrence risk for the next pregnancy as a headline output. The AI interpretation engine summarises the finding in narrative form for clinical letters.
Evagene's batch risk screening flags families whose pedigree structure crosses a configurable mosaicism posterior threshold — useful in large clinical services that want to surface mosaicism candidates automatically from the cohort rather than catching them case by case. The MCP server exposes the mosaicism calculation as a tool so an AI agent can be asked "what is the recurrence risk for the next pregnancy in this family" from inside Claude Desktop or Claude Code.
What competitors do (and don't)
As of April 2026, none of the major clinical pedigree platforms advertises a germline mosaicism posterior calculation on their public marketing pages. Phenotips, Progeny, TrakGene, FamGenix and CanRisk describe cancer risk models and family-history capture; none enumerate mosaicism as a calculated output. This is not a claim that those tools cannot do mosaicism reasoning — enterprise features behind sales may include it — but there is no public feature page for it. Phenotips vs Evagene, Progeny vs Evagene, TrakGene vs Evagene, and FamGenix vs Evagene list mosaicism posterior calculation among Evagene's differentiated capabilities.
The BayesMendel models (BRCAPRO, MMRpro, PancPRO) are carrier-probability models for specific cancer genes and do not model germline mosaicism. CanRisk / BOADICEA is a breast/ovarian/prostate risk model and, similarly, is not a mosaicism calculator. For the dominant and X-linked-dominant non-cancer disorders where mosaicism is clinically dominant — OI, DMD, achondroplasia, TSC, Dravet, Rett — no published web calculator we are aware of currently fills this gap.
Frequently asked questions
What is germline mosaicism?
The presence of a pathogenic variant in some but not all of an individual's germline cells, without being detectable in clinically routine somatic tissue. A mosaic parent is phenotypically unaffected and blood-negative but can transmit the variant to a substantial fraction of offspring.
When should germline mosaicism be suspected?
Two or more affected children born to unaffected parents with an AD or X-linked dominant condition; a family considering another pregnancy after a de novo lethal disorder; advanced paternal age with an AD condition of strong paternal mutational bias.
How does somatic VAF change the risk calculation?
A measured VAF from blood or sperm pins the mosaic fraction posterior and makes recurrence risk approximately VAF × 0.5 × penetrance for AD disorders.
What is joint-parent logic?
When one parent is confirmed mosaic, the other's posterior collapses toward zero. Without this logic, both parents carry residual co-suspicion and counselling is distorted.
How is germline mosaicism different from de novo?
True de novo affects a single gamete only, carrying near-zero recurrence risk. Germline mosaicism affects a measurable fraction of gametes, carrying non-zero recurrence risk.
Which disorders benefit most from this calculator?
Osteogenesis imperfecta (type II), Duchenne muscular dystrophy, achondroplasia, tuberous sclerosis, Dravet syndrome (SCN1A), Rett syndrome (MECP2), neurofibromatosis type 1, Cornelia de Lange.
Does Evagene require VAF data?
No. Posteriors run on pedigree structure and catalogue priors alone. Adding VAF sharpens the estimate but is not required.
Related reading
- Duchenne muscular dystrophy pedigree guide
- Achondroplasia pedigree guide
- Osteogenesis imperfecta pedigree guide
- Tuberous sclerosis pedigree guide
- Dravet syndrome pedigree guide
- Rett syndrome pedigree guide
- Autosomal dominant inheritance calculator
- X-linked recessive calculator
- Inheritance pattern identifier
- Mendelian inheritance calculator
- Rare disease pedigree software